Despite booming growth, uncertainties remain around value demonstration in cell and gene therapies. Roland Berger outlines the challenges and some proposed solutions


US pharmaceutical pricing at a crossroads
The shift toward regulated drug pricing
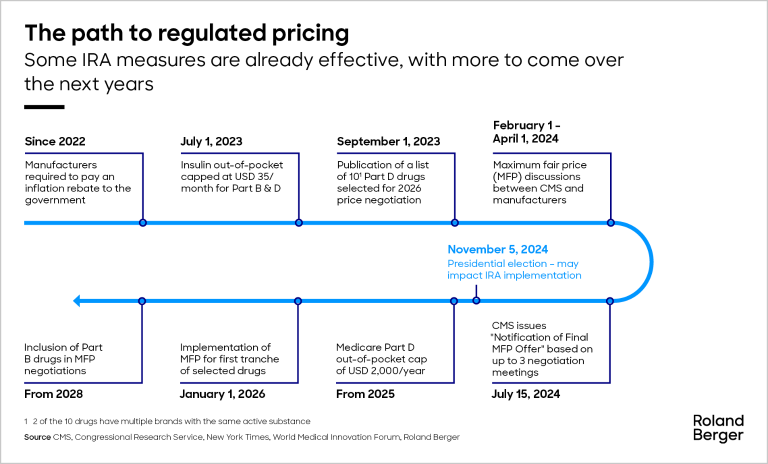
The Inflation Reduction Act means drug price regulation is now a reality in the US. Although still very limited, it is expected to grow rapidly and cause fundamental change in the pharmaceutical market. As regulators announce the first tranche of regulated prices, we look at the implications for companies and patients.

"With health technology assessments and price negotiations now standard in major European markets, US pharma companies could benefit from leveraging European insights as they prepare for price negotiations."
The pharmaceutical market in the United States is currently undergoing its biggest change in more than 20 years. Previously a free-pricing market, price regulation is now being introduced as part of the Inflation Reduction Act (IRA), a multi-billion-dollar stimulus package signed off in 2022. The IRA contains three measures that will result in a paradigm shift in the US pricing and market access landscape – negotiation of drug prices; a redesign of Medicare Part D (the prescription drug benefit for over 65s); and prescription drug inflation rebates. These are expected to result in substantial government savings, estimated at USD 321 billion between 2022 and 2031.
But what will be the impact on patients and pharmaceutical companies? There are many potential implications. For example, while the IRA measures apply only to the Medicare public health insurance program, there is a high risk of spillover into the private sector as Part D Medicare is often managed by private insurers. Or, the capping of some drug prices could push pharma companies to prioritize the development of indications with a smaller proportion of Medicare patients, which are inherently less likely to be exposed to direct negotiations. No matter what happens, companies will need to rethink their strategies.
The Centers for Medicare & Medicaid Services (CMS; the price-regulating body) announced its ‘maximum fair prices’ for the first ten listed drugs on February 1, 2024, triggering a round of negotiations with the industry. To coincide with this, this article explores the key IRA provisions and the shift toward regulated drug pricing, shedding light on their implications for both pharmaceutical companies and patients.
The key IRA measures
Negotiation of drug prices
A core pillar of the IRA is the negotiation of prices on certain high-spend drugs. The first ten drugs have already been chosen, based on Medicare's gross spending and criteria such as the drug having no direct (generic/biosimilar) competition. The federal government must now negotiate directly with manufacturers using a maximum fair price (MFP) offer for each listed drug, calculated using pricing information. The IRA includes tools to compel drug companies to accept the MFPs, including imposing large excise taxes. The first MFPs are set to come into force in 2026.
Medicare Part D redesign
The IRA aims to lower prescription drug premiums for Medicare and involves a significant transformation in the design of Medicare Part D, an enhanced prescription benefit for enrollees. It currently has three phases: initial coverage, with an out-of-pocket (OOP) spending threshold of USD 5,030; the coverage gap, with an OOP threshold of USD 8,000; and catastrophic coverage, for all further spending. The IRA cuts this down to two phases, removing the coverage gap and capping total OOP spending at USD 2,000. In addition, there is an OOP cap of USD 35 on monthly prescriptions for insulin under Medicare Parts D and B (the enrollee’s share of costs).
While these changes come with additional federal spending, they represent substantial financial constraints for pharmaceutical manufacturers and imply a discount on the gross-to-net level of up to 20%.
Prescription drug inflation rebates
Since the IRA was enacted in 2022, manufacturers have been mandated to pay a rebate to the federal government if drug prices increase faster than the rate of inflation. The rebates aim to support Medicare beneficiaries by reducing Part B co-insurance costs. To do this, the government will monitor drug pricing for Part B and D to check if a drug’s price rose faster than the inflation rate. If it does, the manufacturer must pay a rebate to Medicare which will use the money to offset recipients’ Part B payments. For perspective, this mechanism led to the adjustment of co-insurance rates on 47 drugs in 2023.
The implications
The implications of the IRA and the US healthcare system’s transitions toward regulated drug pricing are vast and complex. First and foremost, pharmaceutical companies must adapt to the new negotiating landscape and reconsider their strategies. Here significant insights can be gained from the approaches currently deployed by European pricing authorities.
In theory, the IRA has no impact on the prices paid for medications taken by those who are privately insured. However, in practice, it may open the door for lower prices in the private market, too. For example, in 2023, Eli Lilly lowered the cost of its most prescribed insulins (Humalog and Humulin) by 70% and capped insulin patient OOP to USD 35, regardless of whether they are Medicare enrollees or not. Novo Nordisk (NovoLog, Novolin, and Levemir) and Sanofi (Lantus) have followed a similar strategy.
Furthermore, when the MFPs are announced, it will give pharmacy benefit managers (PBMs) leverage to ask for the same discount, especially in drug classes where there are many other options.
From a health policy perspective, the IRA creates new incentives, not necessarily to the benefit of patients. Notably, the legislation discourages the development of small molecule drugs and encourages the development of large molecule drugs/biologics, as small molecules drugs are eligible to be listed after only seven years on the market, while large molecule drugs/biologics are eligible after 11 years. This could lead to the reprioritization of drug development pipelines for more expensive drugs, to protect margins.
Additionally, pharmaceutical companies may prioritize orphan and single-indication products, which are excluded from the IRA measures. Lastly, manufacturers may refrain from investing in new formulations or delivery method, which are typically introduced in the later stages of the product lifecycle (for example, syringe vs. pre-filled syringe or injection pen).
To complicate matters, the introduction of the IRA measures comes as the FDA has approved drug imports for the first time – Florida has been allowed to import drugs from Canada in an effort to reduce costs. The move compounds the direction of travel in the US to reduce drug costs.
The IRA signifies a paradigm shift for pharmaceutical companies. As the industry adapts to the new negotiating dynamics, strategic considerations become paramount. This presents an opportunity for companies to shape their future strategies in alignment with the evolving healthcare system. Roland Berger stands ready to help them.
We would like to thank Melissa Coyle, Simon Datzer and Elena Muggenthaler for their contributions to this article.
Register now to access the full study, to learn about the challenges, implications and strategies in the post-IRA era. Furthermore, you get regular news and updates directly in your inbox.